Quantifying Patient Experience in Clinical Trials to Drive Continuous Improvement
TrialMark clinical trial services are pioneering the measurement of patient experience in clinical trials through the Patient Trial Experience Scale (PTES). Through an industry coalition led by PatientsLikeMe, and with industry leaders such as AstraZeneca and Takeda, this scale will be presented to patients at study start, mid-point and closeout, simultaneously reflecting ongoing patient feedback while generating data useful for improving future trial design and execution
We aim to transform the industry by spurring an open-source, patient-centered and vendor-neutral trial quality measure. To facilitate this vision, the PTES is open-access. Use of this measure will provide focus to patient-centricity initiatives, allow these initiatives to be scored against a baseline and eventually allow sponsors to compare initiatives, trial types and approaches to patient-centricity. If interested, please contact us to learn how you can adopt the scale in your trial to or advise future phases of this ambitious project.
Contact us to learn how TrialMark will improve clinical trial design through measurement and benchmarking of patient experience
How TrialMark Works

TrialMark has following elements:
- Development of a common, universally applied measure for patient experience in trials that is open-source and free to use; design is driven using robust psychometric principals and heavy patient engagement
- Infrastructure to pool the patient experience data from multiple sponsor companies into a common, de-identified database
- Analysis and tools to help benchmark experiences and derive actionable ideas and best practices for trial design
TrialMark Mission
We aspire that all trials are measured accurately from the patient perspective using a single, openly available, universal tool, and that this data is pooled and disseminated in responsible ways to assist the entire industry to create better trials for patients.

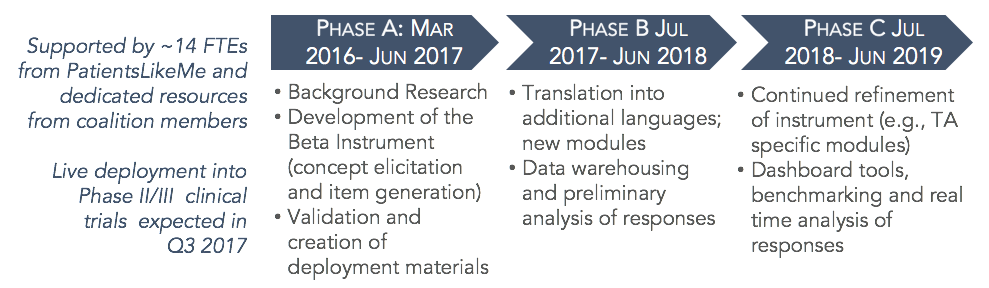
TrialMark Project Roadmap