 Members of our Multiple Sclerosis (MS) community have been discussing this story in the Boston Globe which reports severe adverse events experienced by two European MS patients on the drug Tysabri (Natalizumab). Tysabri is a monoclonal antibody which seems to have a potent effect on MS; the original trial published in the New England Journal of Medicine reported that compared to placebo, MS patients on Tysabri had a 42% lower risk of sustained disability progression over two years, and a 68% reduction in the risk of clinical relapse at one year. Compared to the small and imperceptible benefits from other drugs available at the time, this provided real hope for thousands of patients.
Members of our Multiple Sclerosis (MS) community have been discussing this story in the Boston Globe which reports severe adverse events experienced by two European MS patients on the drug Tysabri (Natalizumab). Tysabri is a monoclonal antibody which seems to have a potent effect on MS; the original trial published in the New England Journal of Medicine reported that compared to placebo, MS patients on Tysabri had a 42% lower risk of sustained disability progression over two years, and a 68% reduction in the risk of clinical relapse at one year. Compared to the small and imperceptible benefits from other drugs available at the time, this provided real hope for thousands of patients.
Tysabri was voluntarily withdrawn in 2005, however, after a small number of cases of a rare condition called progressive multifocal leukoencephalopathy (PML). This extremely rare but usually fatal adverse event seemed particularly prone to happen in patients also taking immunosuppressive drugs; part of the standard toolkit in treating MS. It was reintroduced in 2006 but only within a strict protocol to minimize the risk of adverse events.
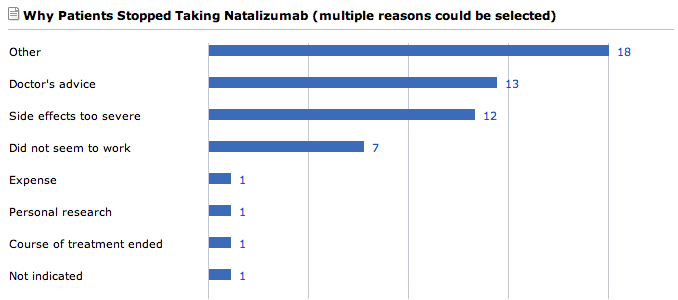
Since the early days of our MS community, Tysabri has been a hot topic of conversation with several patients reporting that their frequency of relapses had dropped substantially and even that some of their symptoms such as bladder dysfunction had improved too. In total, PatientsLikeMe has 297 patients taking Tysabri, although 35 patients stopped taking it, with the most common reasons being doctor’s advice, side effects, or a perceived lack of efficacy. By recording symptoms on their profile pages, our users inform each other about their experiences with Tysabri. Each patient, then is better informed to make their own decision; and by sharing their outcomes they help the whole community to make an informed choice, too.
The one question on the minds of our MS users is “now what”? A disease like MS is hard to understand for those that don’t have it, it’s an “invisible illness” characterized by fatigue, cognitive problems, and pain. When you have symptoms like this impacting upon your quality of life, how can you make the right choice? Given that nothing in our lives is risk free, what is an acceptable balance of risk between relief from suffering for several years and a small risk of death from an adverse event?
Faced with worsening symptoms, empowered patients educate themselves about the potential risks of any treatment; and along with their doctors make the choice that’s right for them. One of our MS members puts it this way:
I understand those who want the PML issue to be understood because it is a very scary thing, but I said to myself that I’d rather fight back with the strongest drug now instead of waiting for that flare up that one day is irreversible.
That is a person who is trying to make the best health care decision based on all of the available information. The choice is yours and we understand it’s not an easy one to make.

Good article Paul. Thanks! Kim
Your headline: “Tysabri And The 2 Patient Deaths Reported” isn’t correct, and is extremely misleading… these two patients are alive, and one is already home while still ambulatory after plasmapheresis (which removes Tysabri from the body), and the other MS patient is currently in the hospital receiving plasmapheresis as well.
It would behoove all current Tysabri patients to get the true and correct facts, rather than rely upon erroneous publications such as Paul’s…. IMHO
Lauren
Lauren,
Thank you for your comment. We have updated the title as it referred to the prior two deaths as well as the newly reported cases of PML where people have not died. For clarity, we have changed the title per your suggestion.
As Paul wrote in his piece, there are almost 300 patients on Tysabri on PatientsLikeMe. These people get information from each other and are well versed in the health care literature. You are a shining example of the type of person who joins PatientsLikeMe.
Thanks again,
David and The PatientsLikeMe Team
Hello, this is my first comment on this post. I also have MS for 25 years and have been on Tysabri for 9 months. I am also a physician! And I too am startled about the two recent reports of PML associated with Tysabri.
Yes there are risks with every treatment vs. its rewards but if the patient’s treatment with Tysabri is handled correctly (I mean monthly blood draws or lumbar punctures to moniter the presence of the virus resposible for PMLs) then I feel there’s nothing to worry about. Because if that virus’s core antigen is detected, then the patient will be taken off the drug and the patients WBCs will force the virus back into dormancy!!
I don’t know if these two people were being monitored or even if they were on any other immunosupressive drugs, such as Copaxone or any of the interfereons. So I don’t know what exactly to make of this news.
Nice site…
So far, only 2 cases of PML have been recorded among Tysabri recipients since the FDA allowed it back on the market…oddly both out of Europe and both announced at the same time. The FDA apparently considers 1 case per 1,000 recipients to be within acceptable risk/reward ratios. Currently, approx 32,000 recip. are receiving Tysabri, with only 2 reported PML cases…that is pretty astounding and demonstrates, at least for now, a much lower incidence than is apparently acceptable. My understanding is that through vigilent screening, PML can usually be caught early and treated successfully; I am unclear re. once PML is treated whether that person can safely go back on Tysabri treatment.
Last year I had two treatments of Tysabri, that was enough for me. Only because the side effects I could not handle.
Which were headaches and alot of pain in my legs. I have to admit that PML did scare me, but my doctor did want me to try Tysabri, but again I could not handle the side effects.
Any drug intervention is unnatural intervention. Tysabri has been linked to 42 cases of brain infection.
CCSVI and it’s surgical correction is a hot topic because it has shown the kind of results no MS drug has achieved.
The financial aspect to MS treatments appears to be hindering the search for a better solution. CCSVI is cheaper and may reduce the need for lifelong inaddequate drugs. Is CCSVI being disregarded because it does not meet the goals of Pharma Company Shareholders. It most certainly looks like it.
Health v Greed