This week on our blog, we’ve been highlighting how patients like you are putting their experiences in context. With the launch of InstantMe and some of the design updates you read about, you can see we’re listening to your call for more functionality that lets you understand how your condition affects the whole you.
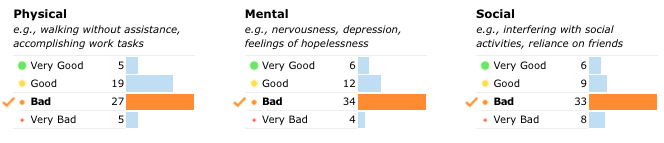
Patient Reported Outcomes (PRO) questionnaires are a great tool to illuminate the physical, mental, and social dimensions of your overall health. In fact, PROs are increasingly used in clinical trials, and in December 2009 the FDA approved the use of PROs to support product claims. Best of all, PROs are free of clinical interpretation, which empowers you, the patient, to have your voice heard in the real world.
PatientsLikeMe is an unparalleled platform for electronic PROs, which have a few advantages over traditional pen-and-paper ones, such as:
- Patients are more likely to share and share truthfully using electronic interfaces;
- Researchers have real-time access to the data;
- Electronic PROs enable alerts for specific concerns (such as adverse side effects), ensuring better safety for all patients (1).
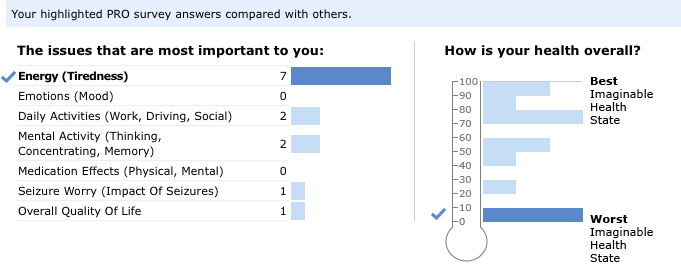
Early on in our partnership, our colleagues at the biopharmaceutical company UCB proposed a longitudinal PRO survey: members of our Epilepsy Community would have access to surveys that reveal the effect of their condition on everyday life over distinct periods. That’s right — you could see your progress over time! Together with UCB, we decided to take the concept of an electronic PRO one step further: not only would you be able better understand your own experience with epilepsy, but you would also be able to put it in context. When you complete a PRO survey, you’ll see that you can compare your results to the larger Epilepsy Community!
Figure 1
Figure 2
Such commitment to patient health reveals two interesting things about UCB: First, patients really do come first; and secondly, UCB is committed to understanding how their products work in the real world, and not just in clinical trials. It is not easy to take a leadership position in an innovative arena such as this, and we are proud to partner with such a company.
 As of today, members of our Epilepsy Community have completed more than 1,300 PRO surveys. By the end of 2010, many of you will have contributed to the largest patient-led study ever in epilepsy.
As of today, members of our Epilepsy Community have completed more than 1,300 PRO surveys. By the end of 2010, many of you will have contributed to the largest patient-led study ever in epilepsy.
Are you in on what our communities already know? Share…and then compare. If you understand your condition in the broader context, you’ll be a PRO too.
1. Dupont et al. “Use of Tablet Personal Computers for Sensitive Patient-Reported Information.” Journal of Supportive Oncology. Volume 7, Number 3. May/June 2009.

Not sure what PRO is but I keep track of my symptoms, and a diary of medication use.