 I came across this Wall Street Journal article earlier this week which details how patients with life-changing illnesses are using online services such as EmergingMed to help them enroll in clinical trials. The article points out that only 3% of adult cancer patients participate in trials, citing lack of awareness as a crucial factor. They write:
I came across this Wall Street Journal article earlier this week which details how patients with life-changing illnesses are using online services such as EmergingMed to help them enroll in clinical trials. The article points out that only 3% of adult cancer patients participate in trials, citing lack of awareness as a crucial factor. They write:
“studies show that the more likely culprit is ignorance… 85% of cancer patients were either unaware or unsure at the time of their diagnosis that participation in clinical trials was an option.”
In addition, there are also systematic flaws in the disjointed way that trials take place which makes it difficult to get accepted into a trial and can make participation unsatisfying even if you do. Say you’ve got ALS (Lou Gehrig’s disease or Motor Neurone Disease), a progressive and incurable life-changing illness and you’re being treated at a specialist centre that runs a lot of clinical trials. Here’s a few scenarios that could happen:
1. The Drug X trial is full before you can participate. Even if you’re suitable for a trial, it’s a matter of chance as to whether you’ll be seen at the hospital during their recruitment window. National, online databases which store shared medical data prospectively could help ensure a fairer system which gave equal opportunity to eligible patients.
2. The Drug Y trial is only looking to recruit a specific subset of patients so you’re not eligible. Maybe you’re eligible for a trial in the next state and they’re desperate for more participants; national trial registries would open up access and make it easier for researchers to recruit all types of patients quickly.
3. The Drug Z trial is an existing drug being used off-label. After taking the drug for 12 months your rate of progression has been slowed significantly and you wish to continue taking the drug. Although the study gets published in an academic journal, it’s not taken that seriously because it wasn’t a double-blind randomized control trial. If patients are willing to continue taking a treatment off-label with the agreement of their primary care physician, the use of an outcomes-sharing site like PatientsLikeMe provides an ongoing opportunity to monitor adverse events and perhaps even evaluate efficacy at little cost (bearing in mind the caveats and biases of such an approach).
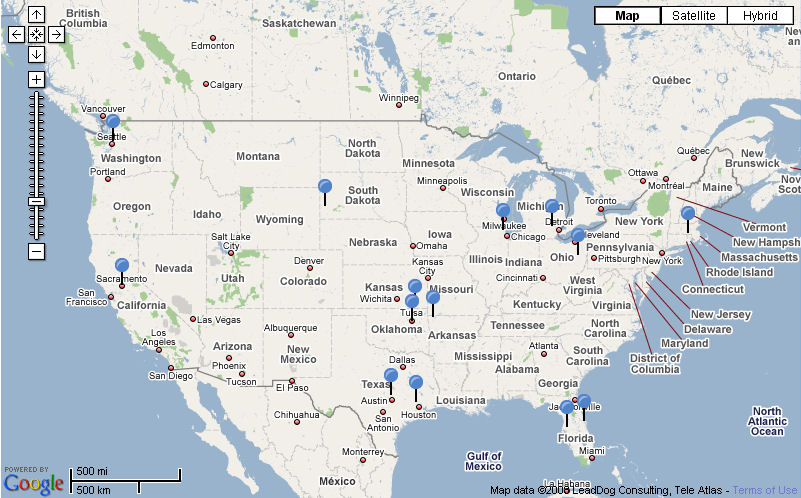 Some members of the medical establishment might say “Well, patients don’t really have a choice. Take it or leave it”. But that’s no longer the case. As our project charting off-label use of lithium in ALS shows, patients are increasingly taking control of their own personal research. Patients are also going the distance to take part in clinical trials they feel offer them the most hope. Our map of ALS patients with a Synapse diaphragm pacing implant shows the distances they have traveled to have the experimental procedure at Case Western in Cleveland, Ohio.
Some members of the medical establishment might say “Well, patients don’t really have a choice. Take it or leave it”. But that’s no longer the case. As our project charting off-label use of lithium in ALS shows, patients are increasingly taking control of their own personal research. Patients are also going the distance to take part in clinical trials they feel offer them the most hope. Our map of ALS patients with a Synapse diaphragm pacing implant shows the distances they have traveled to have the experimental procedure at Case Western in Cleveland, Ohio.
In order to accomplish their goals, clinical trial recruitment will have to change. PatientsLikeMe is playing an active role in driving these changes through from the bottom-up by encouraging patients to share information and take control of their own management. By partnering with organizations running nationwide clinical trials, we want to make it so that clinical research is something you do jointly with patients, not to them.

Dr Paul Wicks,
I am not familiar with the ALS drug trial mentioned in your article, or the cancer trials but it sounds like they, or at least their “fall-out” (outcome) was similar to the GDNF/Amgen negative response to the patient participants such as myself.
I very much appreciated your article and would like to
refer you to the PPP aka http://www.pdpipeline.org/
just in case you were not aware of its exsistence.
sincerely
jingle